3. 结构
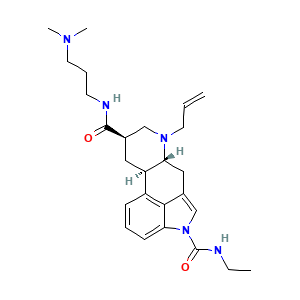
3.1 二维结构
3.2 三维结构
-1
-2
-3
70 73 0 1 0 0 0 0 0999 V2000
-3.2593 -2.3253 1.0693 O 0 0 0 0 0 0 0 0 0 0 0 0
6.7855 -0.8211 0.6442 O 0 0 0 0 0 0 0 0 0 0 0 0
-1.1124 1.2848 -0.5738 N 0 0 1 0 0 0 0 0 0 0 0 0
4.8088 0.2193 0.1742 N 0 0 0 0 0 0 0 0 0 0 0 0
-4.4989 -1.6215 -0.7713 N 0 0 0 0 0 0 0 0 0 0 0 0
6.7408 1.5024 0.4697 N 0 0 0 0 0 0 0 0 0 0 0 0
-7.9135 0.7127 0.6278 N 0 0 0 0 0 0 0 0 0 0 0 0
0.2947 -0.7998 -0.6939 C 0 0 2 0 0 0 0 0 0 0 0 0
0.1814 0.6913 -0.1531 C 0 0 1 0 0 0 0 0 0 0 0 0
-0.9007 -1.6291 -0.2053 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.2122 -0.9413 -0.5690 C 0 0 2 0 0 0 0 0 0 0 0 0
1.4128 1.5696 -0.5901 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.2451 0.4973 -0.0573 C 0 0 0 0 0 0 0 0 0 0 0 0
1.6407 -1.4123 -0.3359 C 0 0 0 0 0 0 0 0 0 0 0 0
2.7225 -0.5297 -0.1880 C 0 0 0 0 0 0 0 0 0 0 0 0
2.6928 0.8664 -0.2914 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.2671 2.6780 -0.1448 C 0 0 0 0 0 0 0 0 0 0 0 0
4.0163 -0.9399 0.0961 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.3634 -1.7010 0.0167 C 0 0 0 0 0 0 0 0 0 0 0 0
1.9277 -2.7757 -0.2143 C 0 0 0 0 0 0 0 0 0 0 0 0
3.9728 1.3101 -0.0707 C 0 0 0 0 0 0 0 0 0 0 0 0
4.2885 -2.3092 0.2400 C 0 0 0 0 0 0 0 0 0 0 0 0
3.2249 -3.2105 0.0760 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.5407 3.2754 -0.6591 C 0 0 0 0 0 0 0 0 0 0 0 0
6.1958 0.2331 0.4508 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.7392 -2.2850 -0.4400 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.5172 3.7445 0.1259 C 0 0 0 0 0 0 0 0 0 0 0 0
-6.6601 -1.4136 0.4119 C 0 0 0 0 0 0 0 0 0 0 0 0
-7.0463 -0.0804 -0.2349 C 0 0 0 0 0 0 0 0 0 0 0 0
8.1469 1.7004 0.7377 C 0 0 0 0 0 0 0 0 0 0 0 0
8.9885 1.4378 -0.4939 C 0 0 0 0 0 0 0 0 0 0 0 0
-8.6268 1.7271 -0.1504 C 0 0 0 0 0 0 0 0 0 0 0 0
-7.1386 1.3455 1.6968 C 0 0 0 0 0 0 0 0 0 0 0 0
0.2581 -0.7630 -1.7930 H 0 0 0 0 0 0 0 0 0 0 0 0
0.2052 0.6443 0.9479 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.8901 -2.6304 -0.6534 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.8215 -1.7745 0.8808 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.3023 -0.9372 -1.6643 H 0 0 0 0 0 0 0 0 0 0 0 0
1.4176 2.5236 -0.0517 H 0 0 0 0 0 0 0 0 0 0 0 0
1.3608 1.7813 -1.6652 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.2047 0.9159 -0.3820 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.2567 0.5090 1.0422 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.4735 3.3034 -0.5663 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.2004 2.7629 0.9477 H 0 0 0 0 0 0 0 0 0 0 0 0
1.1505 -3.5227 -0.3466 H 0 0 0 0 0 0 0 0 0 0 0 0
4.3392 2.3258 -0.0693 H 0 0 0 0 0 0 0 0 0 0 0 0
5.2800 -2.6864 0.4612 H 0 0 0 0 0 0 0 0 0 0 0 0
3.4178 -4.2768 0.1718 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.6688 3.3347 -1.7374 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.4789 -1.0656 -1.6212 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.5154 -3.2166 0.0911 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.2293 -2.5436 -1.3845 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.4161 4.1677 -0.3089 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.4410 3.7119 1.2070 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.1562 -1.2379 1.3692 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.5696 -1.9852 0.6365 H 0 0 0 0 0 0 0 0 0 0 0 0
6.1971 2.3367 0.2925 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.5741 -0.3257 -1.1664 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.1556 0.4899 -0.5283 H 0 0 0 0 0 0 0 0 0 0 0 0
8.2750 2.7348 1.0718 H 0 0 0 0 0 0 0 0 0 0 0 0
8.4526 1.0354 1.5526 H 0 0 0 0 0 0 0 0 0 0 0 0
10.0458 1.6211 -0.2792 H 0 0 0 0 0 0 0 0 0 0 0 0
8.8855 0.4028 -0.8358 H 0 0 0 0 0 0 0 0 0 0 0 0
8.6882 2.0916 -1.3196 H 0 0 0 0 0 0 0 0 0 0 0 0
-9.2574 1.2567 -0.9132 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.9413 2.4228 -0.6480 H 0 0 0 0 0 0 0 0 0 0 0 0
-9.2982 2.3055 0.4941 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.6797 0.6059 2.3595 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.7946 1.9506 2.3336 H 0 0 0 0 0 0 0 0 0 0 0 0
-6.3500 1.9998 1.3070 H 0 0 0 0 0 0 0 0 0 0 0 0
1 19 2 0 0 0 0
2 25 2 0 0 0 0
3 9 1 0 0 0 0
3 13 1 0 0 0 0
3 17 1 0 0 0 0
4 18 1 0 0 0 0
4 21 1 0 0 0 0
4 25 1 0 0 0 0
5 19 1 0 0 0 0
5 26 1 0 0 0 0
5 50 1 0 0 0 0
6 25 1 0 0 0 0
6 30 1 0 0 0 0
6 57 1 0 0 0 0
7 29 1 0 0 0 0
7 32 1 0 0 0 0
7 33 1 0 0 0 0
8 9 1 0 0 0 0
8 10 1 0 0 0 0
8 14 1 0 0 0 0
8 34 1 0 0 0 0
9 12 1 0 0 0 0
9 35 1 0 0 0 0
10 11 1 0 0 0 0
10 36 1 0 0 0 0
10 37 1 0 0 0 0
11 13 1 0 0 0 0
11 19 1 0 0 0 0
11 38 1 0 0 0 0
12 16 1 0 0 0 0
12 39 1 0 0 0 0
12 40 1 0 0 0 0
13 41 1 0 0 0 0
13 42 1 0 0 0 0
14 15 2 0 0 0 0
14 20 1 0 0 0 0
15 16 1 0 0 0 0
15 18 1 0 0 0 0
16 21 2 0 0 0 0
17 24 1 0 0 0 0
17 43 1 0 0 0 0
17 44 1 0 0 0 0
18 22 2 0 0 0 0
20 23 2 0 0 0 0
20 45 1 0 0 0 0
21 46 1 0 0 0 0
22 23 1 0 0 0 0
22 47 1 0 0 0 0
23 48 1 0 0 0 0
24 27 2 0 0 0 0
24 49 1 0 0 0 0
26 28 1 0 0 0 0
26 51 1 0 0 0 0
26 52 1 0 0 0 0
27 53 1 0 0 0 0
27 54 1 0 0 0 0
28 29 1 0 0 0 0
28 55 1 0 0 0 0
28 56 1 0 0 0 0
29 58 1 0 0 0 0
29 59 1 0 0 0 0
30 31 1 0 0 0 0
30 60 1 0 0 0 0
30 61 1 0 0 0 0
31 62 1 0 0 0 0
31 63 1 0 0 0 0
31 64 1 0 0 0 0
32 65 1 0 0 0 0
32 66 1 0 0 0 0
32 67 1 0 0 0 0
33 68 1 0 0 0 0
33 69 1 0 0 0 0
33 70 1 0 0 0 0
4. 国际命名与标识
4.1 IUPAC Name
(6aR,9R,10aR)-9-N-[3-(dimethylamino)propyl]-4-N-ethyl-7-prop-2-enyl-6,6a,8,9,10,10a-hexahydroindolo[4,3-fg]quinoline-4,9-dicarboxamide
4.2 InChl
InChI=1S/C26H37N5O2/c1-5-12-30-16-19(25(32)28-11-8-13-29(3)4)14-21-20-9-7-10-22-24(20)18(15-23(21)30)17-31(22)26(33)27-6-2/h5,7,9-10,17,19,21,23H,1,6,8,11-16H2,2-4H3,(H,27,33)(H,28,32)/t19-,21-,23-/m1/s1
4.3 InChlKey
IRQIOIBAVPLXLF-KJXAQDMKSA-N
4.4 Canonical SMILES
CCNC(=O)N1C=C2CC3C(CC(CN3CC=C)C(=O)NCCCN(C)C)C4=C2C1=CC=C4
4.5 lsomeric SMILES
CCNC(=O)N1C=C2C[C@@H]3[C@H](C[C@H](CN3CC=C)C(=O)NCCCN(C)C)C4=C2C1=CC=C4
4.6 SDF文件
5. 波谱数据
5.1 13C核磁共振谱(13C NMR)
5.2 1H核磁共振谱(1H NMR)
5.3 质谱(MS)
5.4 红外光谱(IR)
5.5 紫外/可见光谱(UV/Vis)
6. 相关药材
7. 相关靶点
8. 相关疾病